
Chemical Reactions of Alcohols, Phenols and Ethers
We all have had hard drinks. Yes, it is alcohol. But apart from human consumption it is used in many other applications or to get different compounds. Similar is with phenol and ether. These chemical compounds (alcohol, phenol, and ether) are reacted with some other compounds and converted to some other chemical. So before beating around let’s get to the point. Below given reactions will let you see these three chemicals from a different perspective.
Suggested Videos
What is Alcohol?
Alcohol is an organic compound which contains a hydroxyl functional group attached to a carbon. We get alcohol from alkenes, carbonyl compounds, hydrolysis of alkyl halides, primary amines, alcohol fermentation, and hydrolysis of ethers.
Browse more Topics under Alcohols Phenols And Ethers
- Chemical Reactions of Ethers
- Introduction and Classification of Alcohols, Phenols & Ethers
- Nomenclature
- Physical Properties of Alcohols, Phenols and Ethers
- Preparation of Alcohols
- Physical Properties of Ethers
- Preparation of Ethers
- Preparation of Phenols
- Some Commercially Important Alcohols
Reactions of Alcohols
Reaction with Metal
When ethanol reacts with sodium metal (a base) sodium ethoxide and hydrogen gas is produced.
2ROH + Na→2RO+Na- + H2
Formation of Halides
Halogens such as chlorine or bromine replace the -OH group in an alcohol.
ROH+ Zn+HCl → R-Cl
R2C-OH alcohol + HCl→ R2CCl
Reaction with HNO3
There is oxidation, accompanied by gas evolution (slow but progressive) in this reaction.
R-OH + HO-NO2→ R-O-NO2
Reaction with Carboxylic Acid (Esterification)
The reaction of the carboxylic acid with an alcohol and an acid catalyst leads to the formation of ester (along with water). This is Fischer esterification.
R-OH +R’-COOH +H+↔ R’-COOR
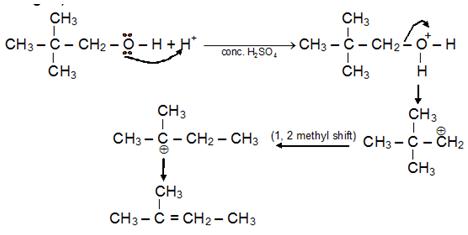
Dehydration of Alcohol
Alcohols dehydrate in an acidic medium. As per the Satyzeff’s Rule, intra-molecular dehydration leads to the formation of alkene while intermolecular dehydration forms ether.
Haloform Reaction
Compound that has the CH3CO- group (or compound on oxidation gives CH3CO - group) which is bonded with a C or H, in the presence of halogen and mild alkali gives haloform. CH3-CH2-COCH2-CH3, CH3-CO-Cl, CH3COOH will not respond to haloform reaction while CH3CH2OH will respond to the haloform reaction.
What is Phenol?
In this chemical compound, a hydroxyl group directly attaches to an aromatic hydrocarbon. Cumene, diazonium salts, etc. form phenols.
Reactions of Phenol
Formation of Ester
Phenyl esters (RCOOAr) do not form directly from RCOOH, but for this acid chlorides or anhydrides react with ArOH in the presence of a strong base.
(CH3CO)2O + C6H5OH + NaOH → CH3COOC6H5 + CH3COONa + H2O Phenylacetate
C6H5COCl + C6H5OH + NaOH → C6H5COOC6H5 + Na+Cl- + H2O Phenyl benzoate
Hydrogenation
Hydrogenation of phenol forms cyclohexanone.
Oxidation of Quinones
Phenols get easily oxidized to para-benzoquinone. This when reduced forms quinones.
Electrophilic Substitution
The —OH and even the —O(phenoxide) are strongly activating ortho, para - directing. Electrophilic monosubstitution in phenols happens in special mild conditions because they are highly reactive and favors both polysubstitution and oxidation.
Halogenation
There is a formation of monobromophenol, on treating phenols with bromine in the presence of a solvent of low polarity like CHCl3 at low temperature.
You can download Alcohols, Phenols and Ethers Cheat Sheet by clicking on the download button below

What is Ether?
Ether is an organic compound that has an oxygen atom, connected to two alkyl and aryl groups, known as the ether group.
Reactions of Ether
Contact with air
When in contact with air, most aliphatic ethers convert to unstable peroxides slowly. The formation of a red color indicates the presence of peroxides. This color appears when the ether is shaken with an aqueous the solution of ferrous ammonium sulfate and potassium thiocyanate.
Halogenation of Ether
Halogenation of ether in dark forms halogenated ethers. Halogens replace the hydrogen atom attached to the C atom which is directly linked to the oxygen atom.
Electrophilic Substitution Reaction
The electrophilic substitution reaction activates the aromatic ring in the same way as in phenol. This is due to the presence of the alkoxy group (−OR) in aromatic ethers activates.
Solved Question For You
Q. What is the difference between alcohol and ether?
Answer: The hydroxyl group of phenol directly bonds to a carbon atom of an aromatic ring, whereas in alcohols, the hydroxyl group attaches to a saturated carbon atom.
Link nội dung: https://sgk.edu.vn/c6h5cooc6h5-naoh-a69683.html